|
|
||||||
|
||||||
|
Beason & Sem: Responses of neurons to
amplitude modulated microwave RF (11/5/02) Authors: Robert C.
Beason1 and Peter Semm2 Authors'
affiliations: Dept. of Biology, State Univ. of New York, Geneseo, NY 15454
USA Present address: 1Dept.
of Biology, Univ. of Louisiana at Monroe, 700 University Ave., Monroe,
LA 71209 USA, 2Fachbereich Biologie, der J. W.
Goethe-Universität Frnakfurt a. M., Seismayerstr. 70, 60054 Frankfurt a.M.,
Germany Corresponding
author: Robert C. Beason, Present address: Dept. of Biology, Univ. of
Louisiana at Monroe, 700 University Ave., Monroe, LA 71209; Telephone:
318-342-1790; Fax: 318-342-3312; E-mail: bibeason@ulm.edu Abstract
In this study we
investigated the effects of a pulsed RF signal similar to the signal produced
by GSM (global system for mobile communication) telephones (900 MHz carrier,
modulated at 217 Hz) on neurons of the avian brain. We found that such
stimulation resulted in changes in the amount of neural activity by more than
half of the brain cells. Most (76%) of the responding cells increased in
their rates of firing by an average 3.5-fold. Other cells responded to the
stimulus with a decrease in spontaneous activity Such responses
indicate a potential health risk for humans using hand-held cellular phones. The postulated biological effects of electromagnetic fields are highly
diverse, ranging from use of natural fields by animals for navigation to
thermal cooking that occurs with strong fields such as produced by microwave
ovens [7]. Athermal effects have been the most difficult to explain because
the mechanism by which they affect biological tissue is usually unknown. It
has been shown that fluctuations of Earth-strength magnetic fields influence
the electrical activity of neurons and pineal cells and the synthesis of
melatonin in birds and mammals [1, 8, 9], including humans [6]. The question
arises as to whether there is a particular sensitivity of the neural tissues
of the brain to high frequency electromagnetic fields such as is produced by
broadcast transmitters. We tested the
effects of electromagnetic radio frequency (RF) signals having a carrier
frequency of 900 MHz, unmodulated and pulse modulated at 217 Hz with a duty
cycle of 12.5% and a power density of 0.1 mW/cm2 because this
signal is similar to that used by the GSM (global system for mobile
communication) telephone system. The test subjects were 34 adult zebra
finches (Taenopygia guttata), anesthetized with a mixture
of ketamine (0.05 mg/g) and xylazine (0.01 mg/g) injected i.m. into the
pectoralis major. The anesthetized bird was mounted in a nonconducting
plastic holder. The bird and the holder were placed inside a tuned RF cavity
(23.5 cm diameter, 100.5 cm long) made of perforated metal. The cavity was
fitted with two tuned RF stubs (each 23.5 cm from opposite ends): one for
emitting the signal and one for monitoring the frequency and power of the
signal within the cavity. To record from neurons in the brain of the bird, a
small hole (4 mm diameter) was made through the skull. A glass
microelectrode (tip diameter 1–2 µm) filled with a conducting solution
physiological saline was slowly advanced into the brain through this hole
until a spontaneously active nerve cell was detected. A silver
reference electrode was inserted beneath the skin along the back of the head
directly behind the glass microelectrode to complete the circuit. Arranging
the electrodes along the long axis of the cavity prevented them from acting
as an antenna and electrically stimulating the cells. Once a spontaneously
active cell was located, it was tested with the stimulus. The protocol
for all the testing procedures was a 10 min prestimulus period, a 10 min
stimulus period, and a 10 min poststimulus period. The rates of the
cell's activity during these three time intervals were compared to detect any
effect of the stimulation. We recorded 133
spontaneously active units from 34 anesthetized adult zebra finches; 91 units
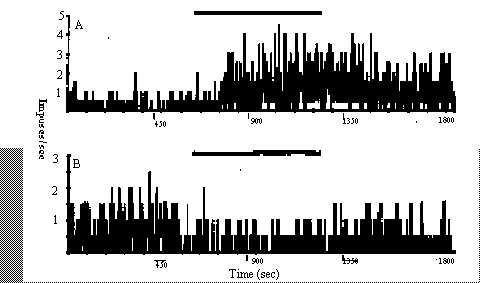
(69%) showed some response to the stimulation: 69 (52%) responded with
excitation (Fig. 1A) and 22 (17%) responded with inhibition (Fig. 1B). The
remaining 42 (31%) cells showed no discernible response. The cells showing
excitation responded with increases in their rate of firing to the
stimulation (mean rate during stimulation = 3.5 ± 0.30 [SE] times prestimulus
rate). Most of the inhibitory responses were small (mean rate during
stimulation = 0.4 ± 0.07 times prestimulus rate), in part because the cells
were firing slowly before the stimulation. Two of the cells showing
inhibition exhibited marked depression in their rates of spontaneous activity
(Fig. 1B). All responses we recorded were to power densities of 0.1
mW/cm2 and stronger (up to 0.5 mW/cm2). The mean
latency from the initiation of the stimulus to the start of the response was
104 (± 197) sec, with the response lasting beyond the end of the stimulus
period in half of the responding cells. The mean persistence beyond the end
of stimulation was 308 (± 68) sec, but there was no correlation (r =
0.489, P > 0.05) between the latency of the response and how long the cell
continued responding beyond the end of the stimulus. Three cells that
responded to the modulated carrier were also tested with an unmodulated
signal of the same carrier frequency. The power of the unmodulated signal was
tested at two densities that equaled the peak power and the average power of
the modulated stimulus. None of these cells exhibited a response to the
unmodulated carrier. In addition to responses to the nominal stimulus, we
also tested four cells that did not respond to the 0.1 mW/cm2
pulsed signal with higher power densities (up to 0.5 mW/cm2).
Three cells did not respond to the stronger intensities, but one cell that
did not respond to the 0.1 mW/cm2 stimulus responded to an
intensity of 0.3 mW/cm2 with depression of its rate of activity. One concern is
that the electrodes themselves were acting as an antenna and stimulating the
cells electrically. The arrangement of the active and reference electrode
along the long axis of the waveguide chamber prevented them from serving as a
loop antenna. In preliminary experiments we varied the positions of the
electrodes to determine whether they could, in fact, act as an antenna.
When the electrodes were not aligned, the stimulus artifact was detected
directly and observed on the oscilloscope display. Whether such a stimulus
was strong enough to stimulate the cells is unknown. A second factor that
supports the idea that the cells were not stimulated electrically is that not all cells responded to the stimulus, even
those in the close neighbourhood of a responding cell. This speaks clearly
against an artifact.. These high
frequency RF fields produced a response in many types of neurons in the avian
Central Nervous System (in both cerebellum and cerebrum) and did not appear to
be limited to any specialized receptor. Similar responses (long latency and ongoing higher activity after
cessation of the fields) also were reported to a 52 GHz carriar, 16Hz modulated signal (Semm er al.,
unpubl. data). Thus, the effect does not appear to be limited to
magnetic sensory cells [10], but may occur in any part of the brain. The
stimulus might mimic a natural mechanism involved in cell communication,
producing responses from many different types of neurons. It is unlikely that
the effects we observed are the result of thermal excitation caused by the RF
radiation because the power densities we applied were 2 to 3 orders of
magnitude below what is required (10 mW/cm2) to produce heating of
even 0.5° C (Bernhardt 1992). Consequently, we conclude that the effects we
observed are not the result of thermal agitation but at this point we cannot
offer an athermal mechanism to account for the observations. Although
individual neurons in the zebra finch brain responded to the pulsed RF
stimulus, we do not know whether these responses by the nervous system are
manifested in the bird's behavior or its health. Bruderer and coworkers [4,
5] reported no behavioral responses of birds to pulsed or continuous RF
microwave signals, but their studies involved different frequencies and lower
power densities of the stimulus. Whether similar neuronal responses occur in
mammals, including humans, requires further investigation. Borbély and
coworkers [3] reported that exposure to a RF signal similar to the one we used
influenced sleep and sleep electroencephalogram in humans. Their results and
the responses we recorded clearly indicate the potential for effects on the
human nervous system. We gratefully
acknowledge financial support of the Deutsche Telekom and the Geneseo
Foundation. Technical assistance and the loan of equipment were provided by
the Deutche Telekom. References
[1] Bartsch, H.,
Bartsch, C., Mecke, D., and Lippert, T. H., Seasonality of pineal melatonin
production in the rat: possible synchronization by the geomagnetic field,
Chronobiol. Int., 11 (1994) 21–26. [2] Bernhardt, J.
H., Non-ionizing radiation safety: radiofrequency radiation, electric and
magnetic fields, Phys. Med. Biol., 37 (1992) 80–84. [3] Borbély, A.
A., Huber, R., Graf, T., Fuchs, B., Gallmann, E., and Achermann, P., Pulsed
high-frequency electromagnetic field affects human sleep and sleep
electroencephalogram, Neurosci. Lett., 275 (1999) 207-210. [4] Bruder, B.,
and Boldt, A., Homing pigeons under radio influence, Naturewissenschaften, 81
(1994) 316–317. [5] Bruderer,
B., Peter, D., and Steuri, T., Behaviour of migrating birds exposed to
x-band radar and a bright light beam, J. Exp. Biol., 202 (1999) 1015–1022. [6] Burch, J.
B., Reif, J. S., and Yost, M. G., Geomagnetic disturbances are
associated with reduced nocturnal excretion of a melatonin metabolite in
humans, Neurosci. Lett., 266 (1999) 209–212. [7] Carpenter, D.
O., and Ayrapetyan, S. [Eds.], Biological Effects of Electric and Magnetic
Fields, Academic, New York, 1994, 2 vols., 726 pp. [8] Reiter, R.
J., Melatonin suppression by static and extremely low frequency
electromagnetic fields: relationship to the reported increased incidence of
cancer, Rev. Environ. Health, 10 (1994) 171–186. [9] Reuss, R.,
and Semm, P., Effects of an earth-strength magnetic field on pineal melatonin
synthesis in pigeons, Naturwissenschaften 74 (1987) 38–39. [10] Semm, P.,
and Beason, R. C., Responses to small magnetic variations by the trigeminal system
of the bobolink, Brain Res. Bull., 25 (1990) 735–740. Fig. 1. Examples of
neuronal responses in the zebra finch brain to stimulation of a 217 Hz, 12.5%
duty cycle square wave modulated 900 MHz carrier signal: A. simulation and B.
inhibition. The solid bar above each graph indicates the presence of the
stimulating RF signal.
|